Understanding how Synovial Joints are formed may help us understand how to influence them to reinitiate longitudinal bone growth.
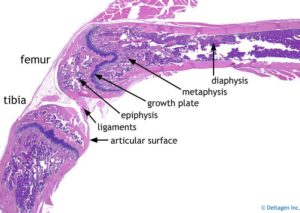
Formation of synovial joints and articular cartilage.
“Chondrocytes differentiate from mesenchymal progenitors and produce templates(anlagen) for the developing bones. Chondrocyte differentiation is controlled by Sox transcription factors. Templates for the neighbour bones are subsequently separated by conversion of differentiated chondrocytes into non-chondrogenic cells and emergence of interzone in which joints cavitation occurs. A central role in initiating synovial joint formation plays Wnt-14/beta-catenin signalling pathway.Moreover, bone morphogenetic proteins and growth and differentiation factors are expressed at the site of joint formation. Joint cavitation is associated with increased hyaluronic acid synthesis. Hyaluronic acid facilitates tissue separation and creation of a functional joint cavity{Hyaluronic acid may inhibit fusion?}. Articular cartilage represents part of cartilage anlage that is not replaced by bone through endochondral ossification. Peri-joint mesenchymal cells take part in interzone formation and that these interzone cells subsequently differentiate into articular chondrocytes and synovial cells. Anlage chondrocytes have a transient character and disappear after cessation of growth plate function while articular chondrocytes have stable and permanent phenotype and function throughout life.”
“Cartilage templates of neighbour bones are separated through development of non-cartilaginous region known as the interzone. The interzone is essential for joint formation since its removal by microdissection led to joint ablation and fusion of bones”<-this is interesting because it means that the interzone is likely a necessary condition for synovial joints and perhaps insertion of the inerzone in other regions can have uniques.
“During interzone formation articular progenitor cell specification prompts the downregulation of the gene for the master chondrogenic transcription factor SOX-9”
“The repressive signals that prevent chondrogenic differentiation within interzone are likely to promote differentiation along a fibrous lineage”
Since Hyaluronic Acid and Heparan Sulfate play a role in cavitation they may help create room for neo-growth plate formation(See Figure 1).
“Once formed, interzone differentiate in 3 layers: 2 chondrogenic, perichondrium-like layers that cover the cartilaginous surfaces and 1 intermediate layer of loose, avascular mesenchyme that separates them and continues peripherally in a vascular mesenchyme, the so-called synovial mesenchyme. Within interzone, along planes destined to become the articular surfaces of synovial joints cavitation occurs”
Necrosis may play a role in preventing cells in the interzone that have chondrogenic potential. Necrosis may be a way that the body blocks neo-growth plate formation.
“HA acid may be an important factor in joint cavitation. Activity of uridine diphosphoglucose dehydrogenase, an enzyme necessary for synthesis of UDP glucuronate, a component for HA production was increased in a narrow band of cells at the presumptive joint line prior to cavitation. Joint cavitation is dependent on the behaviour of fibroblastic cells and/or adjacent chondrocytes, rather than macrophages. Hyaluronan and hyaluronan synthases were present in the interzone before and at the time of cavitation”
” joint cavitation could be facilitated by a rise in local hyaluronan concentration in an area of tissue where cohesion is dependent on the interaction between cellular CD44 and extracellular hyaluronan “<-Since a kind of joint cavitation may be involved to create the region for a neo growth plate, Hyaluronic acid may be helpful in this endeavor.
“The interaction between HA and CD44 can induce both cell adhesion and cell separation, depending on the concentration of HA surrounding a cell population via receptor saturation, with increasing HA concentrations leading to cell separation”<-However, mesenchymal consedation rather than separation is a precursor to growth plate formation.
Genesis and morphogenesis of limb synovial joints and articular cartilage.
“Limb synovial joints are intricate structures composed of articular cartilage, synovial membranes, ligaments and an articular capsule. Together, these tissues give each joint its unique shape, organization and biomechanical function. Articular cartilage itself is rather complex and organized in distinct zones, including the superficial zone that produces lubricants and contains stem/progenitor cells. For many years there has been great interest in deciphering the mechanisms by which the joints form and come to acquire such unique structural features and diversity. Decades ago, classic embryologists discovered that the first overt sign of joint formation at each prescribed limb site was the appearance of a dense and compact population of mesenchymal cells collectively called the interzone. Work carried out since then by several groups has provided evidence that the interzone cells actively participate in joint tissue formation over developmental time. This minireview provides a succinct but comprehensive description of the many important recent advances in this field of research. These include studies using various conditional reporter mice to genetically trace and track the origin, fate and possible function of joint progenitor cells; studies on the involvement and roles in signaling pathways and transcription factors in joint cell determination and functioning; and studies using advanced methods of gene expression analyses to uncover novel genetic determinants of joint formation and diversity. The overall advances are impressive, and the findings are not only of obvious interest and importance but also have major implications in the conception of future translational medicine tools to repair and regenerate defective, overused or aging joints.”
“The superficial zone contains elongated and flat-shaped cells oriented parallel to the articular surface, held together by a largely collagenous matrix, and producing lubricin, hyaluronate and other anti-adhesive macromolecules essential for frictionless joint movement”
“Articular chondrocytes in the middle zone are round in shape, are usually organized in small vertical rows, and produce and maintain important extracellular components – particularly collagen II and aggrecan that confer the tissue with its key biomechanical feature: resilience. The chondrocytes in the bottom zone tend to be larger in size, are also active in matrix production and maintenance, and face the critical tissue boundary – referred to as the tidemark- between articular cartilage and underlying subchondral bone”
“the cartilaginous skeleton forming in the early limb is continuous and uninterrupted, as exemplified by the Y-shaped anlagen corresponding to the stylopod element (humerus or femur), the two zeugopod elements (radius-ulna and tibia-fibula), and the autopod rays corresponding to the phalangeal elements”
” The interzone is composed of flat-shaped cells oriented perpendicularly to the limb’s main axis, tightly bound via gap junctions and requiring the hypoxia regulator Hif-1α function. Because the interzone cells emerge at sites previously occupied by chondrocytes, this led to the suggestion that the interzone cells originate from, and are the direct descendants of, de-differentiated chondrocytes”
“interzone cells are not transient, actively take part in joint tissue formation, and constitute a progenitor cell cohort endowed with joint-formation capacity.”
“rticular and growth plate chondrocytes derive from common mesenchymal precursors originally expressing Dcx which then bifurcate into articular and growth plate chondrocytes. The TGFβ type II receptor (Tgfbr2) is essential for joint formation”
Here’s info on Dcx:
Doublecortin may play a role in defining chondrocyte phenotype.
“Embryonic development of articular cartilage has not been well understood and the role of doublecortin (DCX) in determination of chondrocyte phenotype is unknown. Here, we use a DCX promoter-driven eGFP reporter mouse model to study the dynamic gene expression profiles in mouse embryonic handplates at E12.5 to E13.5 when the condensed mesenchymal cells differentiate into either endochondral chondrocytes or joint interzone cells. Illumina microarray analysis identified a variety of genes that were expressed differentially in the different regions of mouse handplate. The unique expression patterns of many genes were revealed. Cytl1 and 3110032G18RIK were highly expressed in the proximal region of E12.5 handplate and the carpal region of E13.5 handplate, whereas Olfr538, Kctd15, and Cited1 were highly expressed in the distal region of E12.5 and the metacarpal region of E13.5 handplates. There was an increasing gradient of Hrc expression in the proximal to distal direction in E13.5 handplate. Furthermore, when human DCX protein was expressed in human adipose stem cells, collagen II was decreased while aggrecan, matrilin 2, and GDF5 were increased during the 14-day pellet culture. These findings suggest that DCX may play a role in defining chondrocyte phenotype.”
“DCX is expressed in the mesenchymal cells in mouse embryonic limb buds, however, a population of mesenchymal cells maintain DCX expression when they differentiate into joint interzone cells and articular chondrocytes, whereas the other population of mesenchymal cells that differentiate into endochondral chondrocytes lose DCX expression”
“Dlk1 inhibits in vitro chondrogenesis. The increased Dlk1 level may be needed to antagonize other signals that drive chondrogenesis, so as chondrogenesis occurs in a controlled manner”
“Cytl1-null mice also showed normal endochondral ossification and long bone development. In addition, the ultrastructural features of matrix organization and chondrocyte morphology in articular cartilage were similar between wild-type and Cytl1-null mice. However, Cytl1-null mice were more sensitive to osteoarthritic (OA) cartilage destruction upon destabilization of the medial meniscus of mouse knee joints. Furthermore, the expression levels of Cytl1 were markedly decreased in OA cartilage of humans and experimental mice. ”
“Prrx2 was highly expressed in the distal region compared to the proximal region. It has been reported that Prrx2 is highly expressed in undifferentiated mesenchymal cells and its expression decreases when the mesenchymal cells differentiate into chondrocytes ”
“collagen II is expressed at higher levels in endochondral cartilage than articular cartilage”
Toward regeneration of articular cartilage.
“Articular cartilage is classified as permanent hyaline cartilage and has significant differences in structure, extracelluar matrix components, gene expression profile, and mechanical property from transient hyaline cartilage found in the epiphyseal growth plate. In the process of synovial joint development, articular cartilage originates from the interzone, developing at the edge of the cartilaginous anlagen, and establishes zonal structure over time and supports smooth movement of the synovial joint through life. The cascade actions of key regulators, such as Wnts, GDF5, Erg, and PTHLH, coordinate sequential steps of articular cartilage formation. Articular chondrocytes are restrictedly controlled not to differentiate into a hypertrophic stage by autocrine and paracrine factors and extracellular matrix microenvironment, but retain potential to undergo hypertrophy. The basal calcified zone of articular cartilage is connected with subchondral bone, but not invaded by blood vessels nor replaced by bone, which is highly contrasted with the growth plate. Articular cartilage has limited regenerative capacity, but likely possesses and potentially uses intrinsic stem cell source in the superficial layer, Ranvier’s groove, the intra-articular tissues such as synovium and fat pad, and marrow below the subchondral bone. Considering the biological views on articular cartilage, several important points are raised for regeneration of articular cartilage. We should evaluate the nature of regenerated cartilage as permanent hyaline cartilage and not just hyaline cartilage. We should study how a hypertrophic phenotype of transplanted cells can be lastingly suppressed in regenerating tissue. Furthermore, we should develop the methods and reagents to activate recruitment of intrinsic stem/progenitor cells into the damaged site.”
“The growth plate and the chondroepiphysis consisting of the resting zone of cartilage show significant differences in both elastic module and permeability coefficients: the growth plate is half as stiff as the chondroepiphysis and twice as permeable. Permanent hyaline cartilage and transient hyaline cartilage have differences in mechanical property as well as longevity: the former is more stiff and more resistant to compressive force while the latter is more fragile and short lived.”
“The genes expressed in articular cartilage at higher levels include PRG4 (proteoglycan 4), TNC (tenascin C), GREM1 (Gremlin 1), DKK1 (Dickkopf 1), FRZB (Frizzled-related protein 3), ABI3BP (Abl interactor family member), THBS4 (Thrombospondin-4) and SIX1 (sine-oculis-related homeobox transcription factor), while the genes expressed at much lower levels are hypertrophy related genes such as ALPL (tissue-nonspecific alkaline phosphatase), COL10A1 (collagen 10a1) and PTH1R (PTH/PTHLH receptor).”
“Synovial joint formation generally starts with the appearance of a specific population of mesenchymal cells, called interzone cells, at the future joint site between two cartilaginous skeletal elements”
“The interzone cells are characterized by their histological view, a mass packed with elongated cells sandwiched by cartilaginous anlagen and their gene expression profile that is up-regulation of unique molecules such as Wnt-9a, GDF-5, Erg, Gli-3, CD-44, collagen 2A and collagen 1”
“the superficial layer [of articular cartilage] is a pool of stem cells and supports longitudinal and lateral growth of articular cartilage.”
“chondrocytes in permanent cartilage, including articular chondrocytes, retain the potential to proceed toward hypertrophy and that its potential is negatively regulated under the control of the microenvironment that is established and maintained by a fine balance of extracellular matrix components and structure, contents, and activities of various signaling factors. Ectopic hypertrophy of articular chondrocytes is found together with up-regulation of cartilage matrix metabolism in osteoarthritic cartilage”
“Ectopic expression of Wnt9a or activation of its canonical signaling pathway (Wnt/β-catenin signaling pathway) induces ectopic joint-like structure and expression of joint/articular cartilage markers including GDF5”
“Over-activation of TGFβ1 signaling might also accelerate chondrogenic differentiation of stem/progenitor cells in condyle articular cartilage and induce depletion of this cell population. Wnt/β-catenin signaling would maintain stem/progenitor cells in superficial layer, preventing from chondrogenic differentiation.”
“Transient activation of Wnt/β-catenin signaling increases the depth of superficial layer of articular cartilage.”
“joint distraction seems effectively facilitate cartilage regeneration by itself or in combination with a bone marrow stimulation”