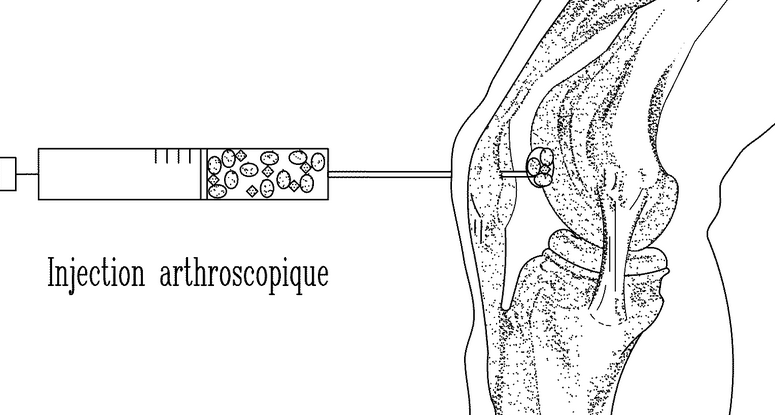
Me: There is two patents I found that has the proposed ideas on how we can possibly inject dna mutated chondrocytes into the epiphysis of the femur and tibia to stimulate mesenchymal stem cells to differentiate into chondrocytes correctly.
Analysis:
For Patent 1 – Only a picture of the idea below was used. The idea as always to combine a type of gel system with growth factors in it and have extracted and regrown chondrocytes implanted in it. Then the entire piece is then implanted into a bone defect or fracture or cartilage to heal it.
For Patent 2 – we have a mixture to heal articular cartilage defects comprising of these components through implantation. There are two main pieces. In essence, it is a proliferating chondrocyte cell structure having phenotypic capability embedded in a vehicle or gel.
The first peice is the extracted and cultured chondrocytes of a certain packing density. This is bone marrow derived chondrocytes or osteoblasts of autologous or homologous origin, or homologous committed chondrocytes, or autologous or homologous muscle fibroblast derived chondrocytes or any other progenital cells from mesenchymal origin
The second part is a a biodegradable, biocompatible, biological resorbable immobilization vehicle (BRIV) adhesive glue. This is made of the following compounds
- fibrinogen (100-150 mg/ml)
- thrombin (60-90 units/ml)
- CaCl₂ (an excess of 60 mM CaCl₂.)
- protease inhibitor (selected from the group consisting of polysaccharide inhibitors, plasma protease inhibitors, and synthetic and natural non-plasma protease inhibitors)
- about 10-30% serum (selected from fetal calf serum or umbilical cord serum from the second trimester or horse serum or any combination of such serums. may optionally contain growth factors such as IGFI, IGFII, TGFB, PDGF, or any other growth factors that will be found to facilitate the proliferation of the cells)
The chondrocyte population is embedded in the biological glue at a concentration of between from 100,000 to 500,000 cells per milliliter of glue.

 For Patent 3 – Patent #3 was written by the same guy who wrote up patent #2 and he wrote it because it turns out patent #2 had a few limitations that was later found out from experiments. It was believed that for the greatest ability in chondrocyte proliferation the best type was embryonic. However this meant the right type of cells was hard to get and there was also the problem with immune system reactions. It would turn out that the concentration limits for the chondrocyte density in gel, the thrombin, and the fibrinogen were not good in actual experimental testing. The compositions and procedures were changed. I have decided just to slip the sections of the patent to the right and left. We can see that overall, the Patent #3 is an improved version to Patent #2 where this has been shown to be more experimentally feasible in causing more chondrocyte proliferation.
For Patent 3 – Patent #3 was written by the same guy who wrote up patent #2 and he wrote it because it turns out patent #2 had a few limitations that was later found out from experiments. It was believed that for the greatest ability in chondrocyte proliferation the best type was embryonic. However this meant the right type of cells was hard to get and there was also the problem with immune system reactions. It would turn out that the concentration limits for the chondrocyte density in gel, the thrombin, and the fibrinogen were not good in actual experimental testing. The compositions and procedures were changed. I have decided just to slip the sections of the patent to the right and left. We can see that overall, the Patent #3 is an improved version to Patent #2 where this has been shown to be more experimentally feasible in causing more chondrocyte proliferation.
Implications: These patents show what exactly would have to go into a gel/resin and cultured cell pellet combination to make any idea of implanted chondrocytes to work out. The way to administer it would be relatively non-invasive through a small syringe with the loaded material going through the knee
Patent #1: Composition and method for the repair and regeneration of cartilage and other tissues (US8258117 B2) (Patent #1 link)
Patent Information:
| Issue No. | US8258117B2 |
| Application Number | 12/901, 293 |
| Public date | September 4, 2012 |
| Filing Date | October 8, 2010 |
| Inventor |
Michael D. Buschmann
2 more » |
| Applicants |
Piramal Healthcare (Canada) Ltd
5 more » |
| United States Patent and Trademark Office patent classification | 514/54 |
Patent #2: Composition for repair of cartilage and bone and method for their preparation as skeletal tissue implant (EP0339607 B1) (Patent #2 link)
A composition for the regeneration of articular cartilage and bones by implantation comprising bone marrow derived chondrocytes or osteoblasts of autologous or homologous origin, or homologous committed chondrocytes, or autologous or homologous muscle fibroblast derived chondrocytes or any other progenital cells from mesenchymal origin and a biodegradable, biocompatible, biological resorbable immobilization vehicle (BRIV) adhesive glue consisiting of fibrinogen, thrombin, CaCl₂, protease inhibitor, and about 10-30% serum, characterized in that said composition contains from 80-160 x 10⁶ cells/ml of BRIV. (From Patent)
Me: Also from the same Inventor Dr. Sam Itay…
Patent #3: Compositions for repair of cartilage and bone (Patent #3 link) [Note: I have already talked about this invention in a previous post]
| A defect is provided in cartilage or bone, or both, to excize damaged or pathological tissue, and it is filled with an implant having capability for complete regeneration of the skeletal tissue as a chondrogenic or osteogenic phenotype. The implant comprises cells expressing a chondrocyte phenotype (80.times.10.sup.6 cells/ml) embedded in a biocompatible matrix having about 20% serum, which provides a permissive environment for maturation and transformation of the implant to a fully integrated state with the surrounding tissue. A portion of the implant may comprise a bone segment or a bone substitute. |