I wanted to spend this post looking at the compositions that are found inside all of the cartilage types, which would include the elastic cartilage, the articular cartilage, and the fibrocartilage. This would help us know what exactly makes up the cartilages that we need to get our bones to lengthen naturally. From doing just a basic google search, I have found two sources which have shown to be very informative.
Note: I will not be restating information that most of us wold already know by now so I will focus on the type of information that are new or insightful
Source #2 at Indiana University – Purdue University Indianapolis: ANAT D502 – Basic Histology – Cartilage, Bone & Joints, Bone Formation Pre-Lab – revised 9.23.12
From Source #1:
For cartilage in general, they seem to have no blood vessels or nerve cells going through them, unlike so many other types of tissue. They are also surrounded by a dense layer of connective tissue known as the perichondrium. it is noted that cartilage in adult humans is very rare. they have the properties of being firm and having the ability to grow rapidly. Cartilage is important in development. They are created when a bone is fractured and is repairing.
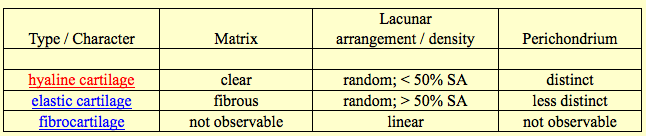
Hyaline Cartilage
So the first type of cartilage that the source looks at is the hyaline cartilage. When a baby before birth still in the fetus form is growing and developing, the precursor cells for chondrocytes, the mesenchymal cells go into a rounded shape and form a densely packed cellular mass called a chondrification centres. There is a chondrocyte forming cell called a chondroblast and they start to secrete the components of the extracellular matrix. There are many substances that are in the extracellular matrix of cartilage but these are the top 4 components.
- Hyaluronan
- Chondroitin sulfate
- Keratan sulfate
- Tropocollagen
The tropocollagen is said to polymerize into collagen fibers that are thin in shape. Whatever the tropocollagen is, the type II of tropocollagen is the dominant form of collagen in all cartilages.
The matrix between the chondroblasts will get bigger and the chondroblasts will separate further from each other in distance. The chondroblasts are located in cavities in the extracellular space of the cartilage, the lucanae. They will also be differentiating into the chondrocytes that we already know about.
Growth – There seems to be two types of growth, interstitial and appositional
- Interstitial growth – Chondroblasts within the existing cartilage divide and form small groups of cells, isogenous groups, which produce matrix to become separated from each other by a thin partition of matrix. Interstitial growth occurs mainly in immature cartilage.
- Appositional growth – Mesenchymal cells surrounding the cartilage in the deep part of the perichondrium (or the chondrogenic layer) differentiate into chondroblasts. Appositional growth occurs also in mature cartilage.
Something interesting that is noted is that the chondrocytes have rough endoplasmatic reticulum which decreases as the immature chondroblasts differentiate into the mature chondrocytes.
Different areas in the cartilage intercellular matrix – There seems to be actually two types of areas in cartilage, territorial and interterritorial matrix. The territorial matrix is located close to the isogenous groups of chondrocytes, which contains larger amounts and different types of glycosaminoglycans.
2 other important things to note:
- Fresh cartilage contains about 75% water which forms a gel with the components of the ground substance.
- Cartilage is nourished by diffusion of gases and nutrients through this gel.
Interestingly, the professor asks in this PDF for the students to “Think about how the spatial arrangement of chondrocytes in the isogenous group may reflect patterns of cell divisions.”
Elastic Cartilage
- occurs in the epiglottic cartilage, the corniculate and cuneiform cartilage of the larynx, the cartilage of the external ear and the auditory tube.
- corresponds histologically to hyaline cartilage, but, in addition, elastic cartilage contains a dense network of delicately branched elastic fibres.
Fibrous Cartilage
- is a form of connective tissue transitional between dense connective tissue and hyaline cartilage. Chondrocytes may lie singly or in pairs, but most often they form short rows between dense bundles of collagen fibres. In contrast to other cartilage types, collagen type I is dominant in fibrous cartilage.
- is typically found in relation to joints (forming intra-articular lips, discs and menisci) and is the main component of the intervertebral discs.
- merges imperceptibly into the neighbouring tissues, typically tendons or articular hyaline cartilage. It is difficult to define the perichondrium because of the fibrous appearance of the cartilage and the gradual transition to surrounding tissue types.
Articular Cartilage
- is a specialised form of hyaline cartilage.
- transforms the articulating ends of the bones into lubricated, wear-proof, slightly compressible surfaces, which exhibit very little friction.
- is not surrounded by a perichondrium and is partly vascularised.
- is, depending on the arrangement of chondrocytes and collagenous fibres, divided into several zones:
- Tangential layer
- Chondrocytes are rather small and flattened parallel to the surface. The most superficial part (lamina splendens) is devoid of cells. Collagen fibres in the matrix of the tangential layer are very fine. They run parallel to the surface of the cartilage.Similar to the collagen fibres of the skin, the general orientation of collagen fibres in articular cartilage is determined by tensile and compressive forces at the articulating surfaces.
- Transitional zone
- The chondrocytes are slightly larger, are round and occur both singly and in isogenous groups. Collagen fibres take an oblique course through the matrix of the transitional zone.
- Radial zone
- Fairly large chondrocytes form radial columns, i.e. the stacks of cells are oriented perpendicular to the articulating surface. The course of the collagen fibres follows the orientation of the chondrocyte columns.
- Calcified cartilage layer
- It rests on the underlying cortex of the bone. The matrix of the calcified cartilage layer stains slightly darker (H&E) than the matrix of the other layers.
The main source of nourishment for articular cartilage is the synovial fluid, which fills the joint cavity. Additional small amounts of nutrients are derived from blood vessels that course through the calcified cartilage close to the bone.
Degeneration and Regeneration of Cartilage
Due to the fairly poor access of nutrients to the chondrocytes they may atrophy in deep parts of thick cartilage. Water content decreases and small cavities arise in the matrix, which often leads to the calcification of the cartilage. This further compromises nutrition. The chondrocytes may eventually die, and the cartilage is gradually transformed into bone.
Chondrogenic activity of the perichondrium is limited to the period of active growth before adulthood. Although chondrocytes are able to produce matrix components throughout life, their production can not keep pace with the repair requirements after acute damage to hyaline or articular cartilage. If these cartilages are injured after the period of active growth, the defects are usually filled by connective tissue or fibrous cartilage. The extracellular matrix of these “repair tissues” is only poorly integrated with the matrix of the damaged cartilage.
Fortunately, cartilage is rather well suited for transplantation – the metabolism of the chondrocytes is rather slow, the antigenic power of cartilage is low, and it is difficult, if not impossible, for antibodies or cells of the immune system to diffuse through the matrix into the cartilage.
From Source #2: