For the longest time I have been hearing from other height increase researchers talk about the Wnt/Beta-Catenin Signaling Pathway and how it effects so many different protein signaling pathways so I wanted to try to explain in my own words in this post what I have come to understand from reading from PubMed articles, Wikipedia articles, Medical journals, and other scientific and biological resources why this pathway is so important and how it is involved in the process of human growth and height.
Since my degree was in chemistry and engineering, I never formally took a class on molecular biology or on genetics before so this will be a slow process. The one class I ever took for Biochemistry I remember almost nothing from it so this study on the Wnt/Beta-Catenin Signaling Pathway will be absolutely necessary for any serious researcher on protein pathways and intracellular and extracellular pathways.
Here is what I remember from high school and college courses.
- The purpose of all genes is to create proteins.
- Genes are found either in the nucleus or the mitochondria of a cell.
- A gene is a molecular unit of heredity of a living organism. It is a name given to some stretches of DNA and RNA that code for a polypeptide or for an RNA chain that has a function in the organism. (Wikipedia)
Since I remember that a lot of biology, biochemistry, and molecular biology, was about remembering names and terms, I think it was important to make a few terms and the terminology clear.
The definitions were mainly taken from the Wikipedia articles on these terms.
- Intracellular – In cell biology, molecular biology and related fields, the word intracellular means “inside the cell”. It is used in contrast to extracellular (outside the cell)….This terms also means existing within the cells.
- Extracellular – In cell biology, molecular biology and related fields, the word extracellular (or sometimes extracellular space) means “outside the cell”. This space is usually taken to be outside the plasma membranes, and occupied by fluid. The term is used in contrast to intracellular (inside the cell).
- Ligand – In biochemistry and pharmacology, a ligand (from the Latin ligandum, binding) is a substance (usually a small molecule), that forms a complex with a biomolecule to serve a biological purpose. In a narrower sense, it is a signal triggering molecule, binding to a site on a target protein.
- Receptor – In the field of biochemistry, a receptor is a molecule most often found on the surface of a cell, which receives chemical signals originating externally from the cell. Through binding to a receptor, these signals direct a cell to do something—for example to divide or die, or to allow certain molecules to enter or exit.Receptors are protein molecules, embedded in either the plasma membrane (cell surface receptors) or the cytoplasm or nucleus (nuclear receptors) of a cell, to which one or more specific kinds of signaling molecules may attach. A molecule which binds (attaches) to a receptor is called a ligand, and may be a peptide (short protein) or other small molecule, such as a neurotransmitter, a hormone, a pharmaceutical drug, or a toxin.
- Cell surface receptors ( aka membrane receptors or transmembrane receptors) – are specialized integral membrane proteins that take part in communication between the cell and the outside world. Extracellular signaling molecules (usually hormones, neurotransmitters, cytokines, growth factors or cell recognition molecules) attach to the receptor, triggering changes in the function of the cell. This process is called signal transduction: The binding initiates a chemical change on the intracellular side of the membrane. In this way the receptors play a unique and important role in cellular communications and signal transduction.
- Kinase – In biochemistry, a kinase is a type of enzyme that transfers phosphate groups from high-energy donor molecules, such as ATP,[2] to specific substrates, a process referred to as phosphorylation.
- Protein – are large biological molecules consisting of one or more chains of amino acids. Proteins perform a vast array of functions within living organisms, including catalyzing metabolic reactions, replicating DNA, responding to stimuli, and transporting molecules from one location to another. Proteins differ from one another primarily in their sequence of amino acids, which is dictated by the nucleotide sequence of their genes, and which usually results infolding of the protein into a specific three-dimensional structure that determines its activity
- Like other biological macromolecules such as polysaccharides and nucleic acids, proteins are essential parts of organisms and participate in virtually every process within cells. Many proteins are enzymes that catalyze biochemical reactions and are vital to metabolism. Proteins also have structural or mechanical functions, such as actin and myosin in muscle and the proteins in the cytoskeleton, which form a system of scaffolding that maintains cell shape. Other proteins are important in cell signaling, immune responses, cell adhesion, and the cell cycle.
- Protein Kinase – is a kinase enzyme that modifies other proteins by chemically adding phosphate groups to them (phosphorylation). Phosphorylation usually results in a functional change of the target protein (substrate) by changing enzyme activity, cellular location, or association with other proteins. The human genome contains about 500 protein kinase genes and they constitute about 2% of all human genes.[1] Protein kinases are also found in bacteria and plants. Up to 30% of all human proteins may be modified by kinase activity, and kinases are known to regulate the majority of cellular pathways, especially those involved in signal transduction.
- Phosphorylation – is the addition of a phosphate (PO43-) group to a protein or other organic molecule. Phosphorylation turns many protein enzymes on and off, thereby altering their function and activity. Protein phosphorylation is one type of post-translational modification.
- Signaling Molecule – is a chemical involved in transmitting information between cells. Such molecules are released from the cell sending the signal, cross over the gap between cells by diffusion, and interact with specific receptors in another cell, triggering a response in that cell by activating a series of enzyme controlled reactions which lead to changes inside the cell.
- Transcription – Transcription is the first step of gene expression, in which a particular segment of DNA is copied into RNA by the enzyme RNA polymerase. Both RNA and DNA are nucleic acids, which use base pairs of nucleotides as a complementary language that can be converted back and forth from DNA to RNA by the action of the correct enzymes. During transcription, a DNA sequence is read by anRNA polymerase, which produces a complementary, antiparallel RNA strand.
From the Wikipedia article on the Wnt Signaling Pathway…
Mechanism
- The Wnt pathway involves a large number of proteins that can regulate the production of Wnt signaling molecules, their interactions with receptors on target cells and the physiological responses of target cells that result from the exposure of cells to the extracellular Wnt ligands.
- The canonical Wnt pathway describes a series of events that occur when Wnt proteins bind to cell-surface receptors of the Frizzled family, causing the receptors to activate Dishevelled family proteins and ultimately resulting in a change in the amount of β-catenin that reaches the nucleus
From the Wikipedia article on Beta-Catenin…
Beta-catenin (or β-catenin) is a protein that in humans is encoded by the CTNNB1 gene. In Drosophila, the homologous protein is called armadillo. β-catenin is a subunit of the cadherin protein complex and acts as an intracellular signal transducer in the Wnt signaling pathway.
Function
β-Catenin is part of a complex of proteins that constitute adherens junctions (AJs). AJs are necessary for the creation and maintenance of epithelial cell layers by regulating cell growth and adhesion between cells. β-Catenin also anchors the actin cytoskeleton and may be responsible for transmitting the contact inhibition signal that causes cells to stop dividing once the epithelial sheet is complete.[6]
Recent evidence suggests that β-catenin plays an important role in various aspects of liver biology including liver development (both embryonic and postnatal), liver regeneration following partial hepatectomy, HGF-induced hepatomegaly, liver zonation, and pathogenesis of liver cancer.[7]
Role in the Wnt signaling pathway
When Wnt is not present, GSK-3 (a kinase) constitutively phosphorylates the β-catenin protein. β-catenin is associated with axin (scaffolding protein) complexed with GSK3 and APC (adenomatous polyposis coli). The creation of said complex acts to substantially increase the phosphorylation of β-catenin by facilitating the action of GSK3. When β-catenin is phosphorylated, it is degraded and, thus, will not build up in the cell to a significant level. When Wnt binds to frizzled (Fz), its receptor, dishevelled (Dsh) is recruited to the membrane. GSK3 is inhibited by the activation of Dsh by Fz. Because of this, β-catenin is permitted to build up in the cytosol and can be subsequently translocated into the nucleus to perform a variety of functions. It can act in conjunction with TCF and LEF to activate specific target genes involved in different processes
The picture below represents a the overview of the major signal transduction pathways. It is taken from the Wikipedia article for signal tranduction pathway .
Note: The post is continued below the picture of the signal transduction pathway diagram. Yes, there is more. Much, MUCH more in this post for me to study and learn. Hope you guys are reading and learning too.
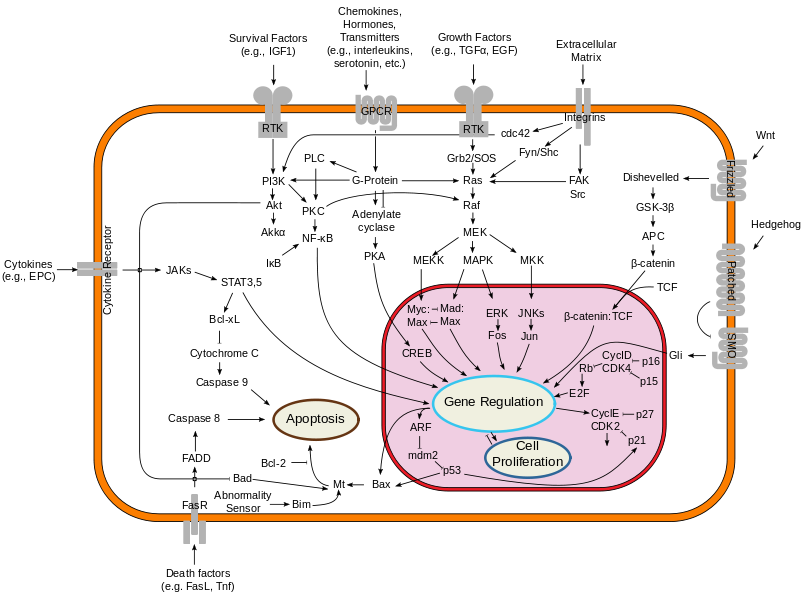
What we are seeing is that the Wnt/Beta-Catenin Signaling Pathway is one of the major signal transduction pathways which goes from the extracellular layer and area through the cell outer membrance inward, into the intracellular area, and then into the Nucleus, through the nuclear membrance.
The first question I would be asking myself is…’What is Wnt? What does the term refer to?”
From what I can gather from the Wikipedia artilce on the word “Wnt” itself, it seems that the term “Wnt” is the combined word and meaning from two words and terms, Int and Wg.
Wg represents Wingless.
Int represents Integrated.
From PubMed study “Wnt signal transduction pathways“…”The name Wnt is resultant from a fusion of the name of the Drosophila segment polarity gene wingless and the name of the vertebrate homolog, integrated or int-1.”
The term Wnt is used in two cases. It refers to both the genes that make a certain type of protein, and the proteins that are made. So there are….
- Wnt genes – The wnt genes make the wnt proteins. (I know, confusing). There is more than one type of Wnt gene, but an entire group (or family) of these genes.
- Wnt proteins – these proteins act almost exclusively as signal transduction proteins, which give signals as a form of communication between individual cells.
From Wikipedia….
- The Wnt proteins are a group of secreted lipid-modified (palmitoylation) signaling proteins of 350-400 amino acids in length
- The WNT gene family consists of structurally related genes that encode secreted signaling proteins. These proteins have been implicated in oncogenesis and in several developmental processes, including regulation of cell fate and patterning during embryogenesis.
- Wnt proteins are a major class of secreted morphogenic ligands of profound importance in establishing the pattern of development in the bodies of all multicellular organisms studied.
From looking at the diagram above, we can assume that the names used in the Wnt/Beta-Catenin Pathway are the key players. There is…
- Wnt –
- Frizzled
- Dihevelled
- GSK-3Beta
- APC
- Beta-Catenin
- TCF
It would be difficult for me to explain the pathway well right now so I take a paragraph from a page from Stanford…
Wnt proteins form a family of highly conserved secreted signaling molecules that regulate cell-to-cell interactions during embryogenesis. Insights into the mechanisms of Wnt action have emerged from several systems: genetics in Drosophila and Caenorhabditis elegans; biochemistry in cell culture and ectopic gene expression in Xenopus embryos. Mutations in Wnt genes or Wnt pathway components lead to specific developmental defects, while various human diseases, including cancer, are caused by abnormal Wnt signaling. As currently understood, Wnt proteins bind to receptors of the Frizzled and LRP families on the cell surface. Through several cytoplasmic relay components, the signal is transduced to beta-catenin, which enters the nucleus and forms a complex with TCF to activate transcription of Wnt target genes
Me: From the Stanford website, there is 19 documented Wnt genes from the mice genome. The reason why we can say that the study of mice to find elementary protein pathways and that information can be eventually applied to humans is because of this. It would appear that the Wnt pathway is a type of signal transduction, extracellularly pathway that almost all animals seem to have, derived from some billion years ago evolved process. If there is 19 currently found Wnt genes, we could be reasonably confident in saying that there is probably 19 Wnt genes for humans as well, and that the genes for humans have the same function as the genes in the mice.
From the looks fo the diagram, it seems that the Wnt gene will create a Wnt protein, which is and acts as a signal transduction from one cell to another cell. It is also a ligand. When it reaches the outer surface of the cell, it interacts with a certain type of receptor. This process is a where the Wnt ligand/protein and attaches itself to a specific type of protein, known as the target protein. The surface of animal cells is called the plasma membrane.
From a first glance, it would seem that the Wnt protein first interacts with something called a Frizzled, which is a type of protein already on the plasma membrane. This send a signal to something called a Dihevelled.
From the webpage from Stanford….
- Wnt –
- Frizzled – Frizzled proteins are seven-transmembrane receptors. There is biochemical and genetic evidence that Frizzleds act as receptors for Wnt proteins, in particular in the case of Frizzled and Dfrizzeld2 interacting with Wingless in Drosophila. Wnts can bind the the CRD (cysteine-rich domain) of Frizzled, an extracellular part of the receptor. The structure of the CRD has been solved by Dann (2001) FRP/FrzB molecules consist of the CRD only and can act as secreted antagonists of Wnt signaling. Little is known about the mechanism of Frizzled signaling. Some but not all Frizzleds stimulate Ca release and PKC activity.
- Dihevelled – Dishevelled is one of the multi-module proteins working in the Wnt pathway. The DIX domain in Axin is similar to a domain in Dishevelled, and may promote interacions between these two domains. The protein has been found at multiple locations in cells, including the nucleus
- GSK-3 – GSK-3 (zw3 or shaggy in Drosophila) is a key enzyme in Wnt signaling. It phosphorylates b-catenin leading to the subsequent degradation of this molecule. The phosphorylation is primed by CK1alpha.
- APC – APC is an enormous protein that has muliple roles in the cell. In the Wnt pathway, it binds to b-catenin and is necessary for its down-regulation. APC also interacts directly with Axin.
- Beta-Catenin – β-catenin (armadillo in Drosophila) is the key mediator of the Wnt signal. In cells not exposed to the signal, β-catenin levels are kept low through interactions with the protein kinase zw3/GSK-3, CK1a, APC and Axin. β-catenin is degraded, after phosphorylation by GSK-3 and CK1 alpha, through the ubiquitin pathway.
- TCF – In the nucleus, in the absence of the Wnt signal,TCF acts as a repressor of Wnt/Wg target genes. TCF can form a complex with Groucho. The repressing effect of Groucho is mediated by interactions with Histone Deacetylases. b-catenin can convert TCF into a transcriptional activator of the same genes that are repressed by TCF alone
Axin – Axin associates directly with β-catenin, GSK-3β and APC and is implicated in down- regulating Wnt signaling. A gene related to Axin, called Conductin or Axil was cloned by virtue of interacting with β-catenin. Overexpressed Axin or Conductin destabilize β-catenin
So the pathway from the diagram goes Wnt –> Frizzled –> Dihevelled –> GSK-3Beta –> APC –> Beta-Catenin –> TCF –> Gene regulation.
From the information I have gathered through Wikipedia and Stanford, this is what I can piece together at this point on the entire pathway.
- The Frizzled is a big receptor that goes across the lipid bilayer which is the plasma membrane. It is big, long, and has ends sticking out to the outside and side the cell. The Frizzeled act as a receptor for the Wnt proteins.
- The GSK-3 is an enzyme in the pathway. It adds a phosphate group to beta-catenin which causes the beta-catenin to degrade. Before it can add the phosphate group, there is another element called CK1alpha which needs to do something on the GSK-3.
- The APC seems to have multiple roles in the cell. For the Wnt pathway, it binds to the beta-catenin to down regulate the beta-catenin. It also interacts directly with Axin.
- The Beta-Catenin itself does not directly interact with the Wnt signal from the beginning of the pathway. The levels of this element is kept low from binding and interacting with other elementts, the GSK-3 (aka zw3), the CK1a, the APC, and the Axin. The Beta will be phosphorylated by the GSK-3 and the Ck1 alpha which will cause it to degrade.
- For the TCF, its function is to repress Wnt/Wg target genes if there is to Wnt signal.
- The Axin is used to down-regulate Wnt signaling.
From this quick analysis on why the Wnt/Beta-Catenin signaling pathway is important for growth and overall height, it would seem that the pathway is one of the most important in controlling the life cycle of cells during embryo formation.
From PubMed study “Wnt signal transduction pathways“…
“The extra-cellular Wnt signal stimulates several intra-cellular signal transduction cascades, including the canonical or Wnt/β-catenin dependent pathway and the non-canonical or β-catenin-independent pathway which can be divided into the Planar Cell Polarity pathway and the Wnt/Ca2+ pathway”
Interpretation: This sentence implies that there Wnt/Beta-Catenin signal pathway is one of several pathways all started by the extracellular Wnt signal. The canonical version has the Beta-Catenin and the non-canonical does not have the beta-catenin. The study would go on to explain why the Wnt family of genes and protein and the pathway named after them are so important in growth and development of the human body….
Conclusion: I wanted to end this post here due to the fact that the Wnt/Beta-Catenin signaling pathway is one of most important and most studied intracellular signaling pathways. There is much more to learn about this pathway than what I have learn here and I think that I should come back to the learning of this pathways in another time. There are quite a few PubMed studies and articles which show how the target genes and the eventually expressed (or not expressed) proteins result in regulating some part of a the chondrocyte proliferation and/or chondrocyte differentiation processes. As for now, there is clear connections on how the Wnt/Beta-Catenin signaling pathways effects growth, but I just haven’t had the chance or have more time to go further into studying and researchers its effects.
 I was reading through the science articles recently when I came through an article which talks about innovative medical innovations on the science of the eye. The BBC, Discovery Magazine, Washington Post, and other sources all have been able to pick up this story of the fact that a optic physician from California, a Dr. Gregg Homer, says that he has managed to create a type of laser that can in only 20 seconds remove the pigmentation from a person’s iris.
I was reading through the science articles recently when I came through an article which talks about innovative medical innovations on the science of the eye. The BBC, Discovery Magazine, Washington Post, and other sources all have been able to pick up this story of the fact that a optic physician from California, a Dr. Gregg Homer, says that he has managed to create a type of laser that can in only 20 seconds remove the pigmentation from a person’s iris.