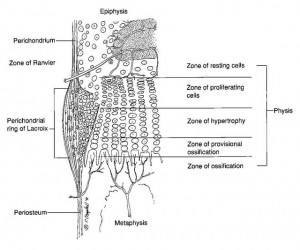
Here’s a diagram showing the zone of Ranvier in relation to the growth plate:
If we know what stresses are responsible for forming growth plates we can re-induce these mechanical stresses to form new growth plates.
This study would be more significant except it was published in 1988 before the scientific community began to focus more on genes and cellular epigenetics. Just because the highly undifferentiated cells of a juvenile or infant may be induced to differentiate into the cells composing the Zone of Ranvier and in turn the growth plate does not mean that adult mesenchymal stem cells will do the same thing.
And I think going back to these old studies is a good thing as they focued more on mechanical factors rather than retrovirus genetic engineering.
One study found that interstitial fluid flow upregulates the mesenchymal condensation gene Msx2 but downregulates the chondrogenic gene Col2a1. This is not necessarily a bad thing as mesenchymal condensation in a neo-Zone of Ranvier would be key to forming new growth plates and those genes would not be chondrogenic until they became a growth plate. In fact in this study the Zone of Ranvier experienced more osteogenic than chondrogenic stimuli so it would be no surprise that a mesenchymal stem cell that may eventually become a growth plate chondrocyte may not express chondrogenic genes initially.
This study provides additional evidence for LSJL as LSJL compresses the bone and the joint thus altering the “Loading history” of the bone and joint. Since the shear strain, caused by LSJL fluid flow, needs to be high this could explain why I’m getting more results for my fingers than my legs.
The role of mechanical loading histories in the development of diarthrodial joints.
“The role of mechanical loading history in chondroosseous development at the ends of long bones is explored using two-dimensional finite element models of chondroepiphyses. Loading histories are characterized in terms of discrete loading cases defined by joint contact pressure distributions and an associated number of loading cycles. An osteogenic stimulus throughout the chondroepiphyses is calculated following the theory that cyclic octahedral shear stresses promote endochondral ossification and cyclic compressive dilatational stresses inhibit ossification{Octahedral stresses are shear stresses that are acting on octahedral planes inside the bone. The plane whose normal vector forms equal angles with the coordinate system is called octahedral plane. But basically shear stress which is what LSJL can induce. A compressive dilational stress is the increase in volume per unit volume of a homogeneous substance. So for example if you squeezed a stress ball and it got bigger}. The resulting distributions for the osteogenic stimulus predict the appearance of the secondary ossific nucleus and the shape of the developing bony epiphysis. The zone of Ranvier and the formation of articular cartilage and the growth plate are predicted by the models{Considering that the zone of Ranvier is the basis of the growth plate this is key}. Tissue stress histories constitute an important influence during skeletal morphogenesis {And we can expose the tissue to different stresses ala LSJL}.”
“Alterations in joint loading or motion can alter the pattern of ossification and growth in developing bone ends, and a reduction in joint forces can delay the appearance of the secondary ossific nuclei”
“The sequence of cartilage proliferation, maturation, degeneration, and ossification is the normal process for all cartilage in the appendicular skeleton. This process is (a) accelerated by intermittently applied deviatoric (shear) stresses (or strain energy) and (b) inhibited or prevented by intermittently applied compressive dilatational stresses (hydrostatic pressure).”
“The appearance of the secondary ossific nuclei in both chondroepiphyses was predicted. Increased osteogenic stimuli were also calculated at the edge of the advancing ossification front where the zone of Ranvier (ossification groove) forms. The areas where the osteogenic stimuli were low define those cartilaginous regions that become the growth plate and the articular cartilage.”
“the ossific nucleus appears in an area of high shear (deviatoric) stresses the edge of the advancing ossification front (zone of Ranvier or ossification groove) also experiences high shear stresses, and the joint surface, where articular cartilage forms, is exposed to high-magnitude hydrostatic compression”<-Since the zone of Ranvier is the key forming new growth plates, inducing high shear stress would be key to forming new growth plates and LSJL can induce shear stress via interstitial fluid flow. According to the study Interstitial fluid flow: the mechanical environment of cells and foundation of meridians., interstitial fluid flow induces shear stresses. Here’s Michael’s summary of LSJL.
“In both the convex and the concave chondroepiphysis, a state of high hydrostatic pressure is created directly beneath the loaded contact surface. In the interior regions of both chondroepiphyses, areas of high-magnitude octahedral shear stress are created. The location of these areas within the central zone of the chondroepiphyses shifts with the direction of the applied loading”<-If we mimic this state we can achieve chondroinduction.
“the shape of the developing bony epiphysis will depend on the geometry of the bone end. In the model with a convex joint surface, the developing ossific nucleus is stimulated to produce a sphere-shaped bony epiphysis. The model with a concave joint surface is stimulated to create a flatter, more disc-shaped bony epiphysis.”
“The stored energy in cartilage under mechanical loading is primarily in the form of deviatoric or shear energy owing to the nearly incompressible nature of this tissue. Some of this stored deviatoric energy is lost in hysteresis upon unloading. The energy dissipated during intermittent mechanical loading must be accounted for by a change in internal energy and/or a change in the temperature of the cartilage. It is possible that the direct mechanical alteration of cells or the increased temperature associated with energy dissipation caused by intermittent shear stresses could increase mitotic activity or activate specific biochemical pathways in the cells”
Here’s another study with some information on the Zone of Ranvier:
Stem Cells and Cartilage Development: Complexities of a Simple Tissue
“[The] biochemical composition [of cartilage] is uniquely suited to providing a combination of tensile strength with deformability, giving it mechanical properties that resemble those of a shock absorber, thereby dissipating forces across the bones, preventing them from fracturing during normal activity.”
“The balance between mechanical stiffness and flexibility is itself the result of interaction between the thin type II collagen fibrils, giving tensile strength, within which are trapped molecules of aggrecan, which are highly negatively charged and so bind water avidly”
“When unloaded, the water content of cartilage is about 70% of the wet weight. Under deforming load water flows out and when the load is reduced it flows back in, damping the effects of these forces. Damage to either the type II collagen or aggrecan may lead to loss of cartilage function”<-Maybe when LSJL loads the cartilage it results in water flowing out and possibly other nutrients and maybe even growth factors that can result in neo-growth plates.
“During cartilage development [cartilage transforms] from a relatively simple isotropic tissue with a high cell density and homogeneous distribution of collagen fibrils to an anisotropic tissue with a low density of chondrocytes growing in vertical columns and a unique arrangement of collagen fibrils.”
“Articular cartilage chondroprogenitor cells are derived from migration of mesenchymal cells out of the zone of Ranvier niche. In early development these cells may accumulate in the surface zone and drive the process of appositional growth of cartilage but with maturity they become dissipated throughout the cartilage.”<-So maybe LSJL can drive these cells back into the zone of ranvier?