Factors affecting bone growth
“Longitudinal bone growth depends on the growth plate{although some other regions may contribute in a minor way}. The growth plate has 5 different zones—each with a different functional role—and is the final target organ for longitudinal growth. Bone length is affected by several systemic, local, and mechanical factors. All these regulation systems control the final length of bones in a complicated way. ”
“Bone growth in width is also controlled by genetic factors, but mechanical loading regulates periosteal apposition”<-periosteal stem cells have the potential to differentiate into chondrocytes so it would be possible to use the mechanical loading to induce new longitudinal bone growth.
“Differences in bone size are established early in life, before puberty and perhaps even in utero. Bone begins to form when mesenchymal cells form condensations—clusters of cells that adhere through expression of adhesion molecules”
“Two models for control of bone growth in width have been suggested—the mechanostat theory (mechanical requirements regulate periosteal apposition) and the sizostat hypothesis (a master gene or set of genes regulates bone growth in width so bone reaches a preprogrammed size, independent of mechanical requirements)”
“The growth plate consists mainly of collagen fibrils, proteoglycans, and water, arranged to form a sort of sponge with very small pores. The growth plate is located between epiphyseal and metaphyseal bone at the distal end of long bones and is strain-rate–dependent, which means it is hard when squeezed rapidly but soft when deformed slowly{the means that the frequency to which stimuli is applied to the growth plate can result in a large variation in response}. The growth plate becomes ossified after puberty and epiphyseal fusion.”
“Histologically, the growth plate consists of horizontal zones of chondrocytes at different stages of differentiation. The germinal zone, at the epiphyseal end of the growth plate, contains resting chondrocytes, which seem crucial in orienting the underlying columns of chondrocytes and, therefore, in unidirectional bone growth, probably by secretion of a growth plate–orienting factor. Next is the proliferative zone, a matrix-rich zone in which flattened chondrocytes undergo longitudinal cell division and orient themselves in typical column-wise fashion. At some point, proliferating chondrocytes lose their capacity to divide; they start to differentiate and become prehypertrophic, coinciding with a size increase. Proliferating chondrocytes are located in the transition (maturation or prehypertrophic) zone. In the hypertrophic zone, round chondrocytes secrete matrix proteins in large amounts. This stage is characterized by an increase in intracellular calcium concentration, which is essential in the production of matrix vesicles. These vesicles, small membrane-enclosed particles, are released from chondrocytes and secrete calcium phosphates, hydroxyapatite, and matrix metalloproteinases, resulting in mineralization of the vesicles and their surrounding matrix. The chondrocytes in this mineralized zone eventually undergo programmed cell death (apoptosis), leaving a scaffold for new bone formation. ”
“Generally, bones increase in length as long as new material is being squeezed between the reserve zone of the growth plate and the zone of provisional calcification.”<-Can we still do this post epiphyseal fusion?
“Intracellular calcium concentration increases in the hypertrophic chondrocytes in the hypertrophic zone of growth plate cartilage; at some point, these chondrocytes begin to mineralize the longitudinal septa in the surrounding matri. At the growth cartilage junction, mononuclear cells of undetermined origin resorb the unmineralized horizontal septa of the growth cartilage. These cells are called septoclasts or chondroclasts. Blood vessels invade the area and pave the way for bone cell precursors. Eighty percent of the longitudinal septa of the growth cartilage is rapidly resorbed in the metaphyseal zone immediately behind the invading blood vessels, paving the way for bone cell precursors. About 40% of mineralized septa serves as scaffold for the formation of primary bone trabeculae; the other 60% is absorbed by chondroclasts (osteoclasts) near the vascular invasion front. ”
The scientists list factors that positively influence bone growth:

I think to or after a certain level should be applied to almost all of these like Estrogen. This list shouldn’t be taken as gospel as there is a lot more detail in how these hormone influence longitudinal bone growth. Innervation is to supply a body part with nerves and I think too has a more complex relationship to longitudinal bone growth than is alluded too.
“GH acts on resting zone chondrocytes and is responsible for local IGF-1 production, which stimulates clonal expansion of proliferating chondrocytes in an autocrine/paracrine manner. Infusion of GH or IGF-1 shortens stem- and proliferating-cell cycle times in the growth plate of hypophysectomized rats and decreases the duration of the hypertrophic differentiation phase, with GH being more effective. According to [an] experimental study, GH or IGF-1 treatment restores mean cell volume and height, but the growth rate is not normalized by either hormone. ”
“hyperthyroidism increases the growth rate in children but also leads to premature growth plate fusion and short stature. T3 seems to stimulate recruitment of cells from the germinal zone to the proliferating zone and facilitates differentiation of growth plate chondrocytes. Its precursor, T4, increases the number of [3H]methylthymidine-labeled chondrocyte nuclei and [35S]incorporation in Snell dwarf mice growth plates, suggesting a stimulatory role in chondrocyte proliferation and differentiation”
However, I think this view overemphasizes the importance of hormones. You can read more on hormones in the full study which I provided a link too in the top.
“If compression always inhibited bone growth, as it was believed, growth plates would be extremely unstable, as any slight deviation from the straight alignment of the long bones of the lower extremities would induce a vicious circle of positive feedback and result in catastrophic deformities. Mild compression leads to increased, not decreased, growth. Nevertheless, when compression on one side of the growth plate exceeds a certain level, growth is indeed suppressed, and the lesion begins to worsen.”
 Hypothesized theory of input of compression and tension on growth rate.
Hypothesized theory of input of compression and tension on growth rate.
“bone cells accommodate to a customary mechanical loading environment, making them less responsive to routine loading signals”
“muscle pull affects periosteal tension and, consequently, bone form and growth in length”
“wider bones must have higher midshaft periosteal apposition rates, as this is how they
become wider.”

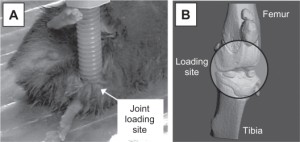
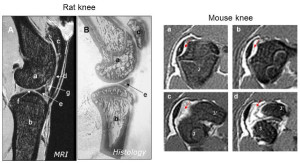
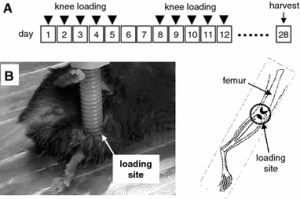
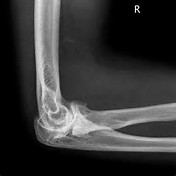


 Hypothesized theory of input of compression and tension on growth rate.
Hypothesized theory of input of compression and tension on growth rate.